If you’ve ever cracked open a cold Perrier between rounds at happy hour, or reached for a Topo Chico to reset your palate between cocktails, you’ve probably noticed it: that sharp, faintly bitter edge that lingers after every sip. It doesn’t taste like still water. It doesn’t taste like soda. It sits somewhere strange, a little acidic, a little metallic, and for many people, genuinely unpleasant at first. So what’s actually going on?
This isn’t just a matter of personal preference. The bitterness in sparkling water has a real chemical origin, a neurological pathway, a genetic dimension, and even a connection to the minerals lurking in your favorite brand’s source spring. If you enjoy a crisp lager, a well-made gin and tonic, or a glass of Champagne on a Friday night, understanding why sparkling water tastes the way it does will actually deepen your appreciation of all three. The science of fizz is more fascinating, and more personal, than you might expect.
You Are Watching: Why Does Sparkling Water Taste Bitter Updated 05/2026

The Core Culprit: Carbonic Acid and What It Does to Your Mouth
The story starts with a simple chemical equation. When carbon dioxide (CO₂) is dissolved into water under pressure, it doesn’t just sit there inertly creating bubbles. It reacts with the water molecules to form carbonic acid (H₂CO₃):
CO₂ + H₂O → H₂CO₃
The carbon dioxide reacts with the water and produces carbonic acid, and this is why sparkling water has a slightly bitter taste. It’s the carbonic acid you are tasting.
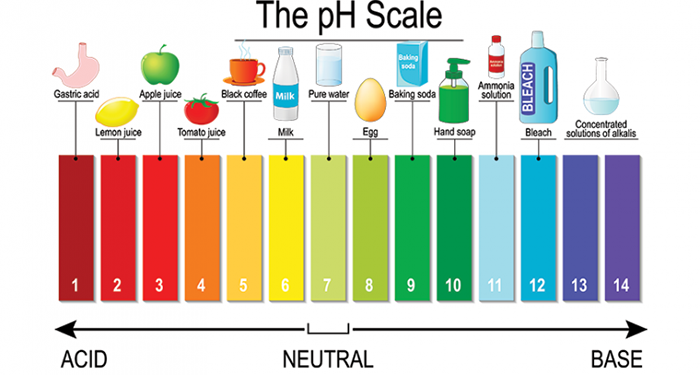
This weak acid lowers the pH from neutral (7.0 for still water) to approximately 4–5, creating that characteristic crispness. That might not sound like much, but your taste receptors are extraordinarily sensitive to even minor pH shifts. The drop is enough to register as distinctly acidic, and often as bitter.
Here’s where it gets more nuanced. CO₂ is a weak acid and gives sparkling water a bitter, citrus-like or sour taste, much like lemon juice which contains citric acid. This is why many people can’t quite put their finger on whether sparkling water tastes sour or bitter. Often, it’s both, depending on your individual biology.
The Bubble Myth That Scientists Had to Debunk
For a long time, people assumed the bitterness came from the bubbles themselves, specifically from the sensation of them popping against the tongue. That theory has since been proven wrong. These studies found that it wasn’t a reaction to the bubbles popping. They found this out when they did studies of people drinking carbonated water that had gone flat. The flat carbonated water still produced the same bitter taste that the carbonated water that still bubbled did.
In other words, even after the fizz dies, the chemical residue of carbonic acid remains in solution, and that’s what your tongue is picking up. The bubbles are the show; the acid is the actual actor.

Your Brain, Your Trigeminal Nerve, and a Hidden Pain Signal
One of the most surprising discoveries in carbonation research is that the “bite” of sparkling water isn’t processed purely through your taste buds. The main mechanism of carbonation sensation is due to CO₂ converted via carbonic anhydrase into carbonic acid, which excites trigeminal receptors and activates the cranial nerve V (the trigeminal nerve). Moreover, the tingling sensation due to CO₂ persists long after the carbonated water has been expectorated.
The trigeminal nerve is the same cranial nerve responsible for pain and sensation across your face. It’s why horseradish makes your eyes water, and why jalapeño burn feels so intense. When carbonic acid forms on your tongue, it activates lingual nociceptors, which in turn excite trigeminal neurons involved in signaling oral irritation.
Yes, technically, every sip of sparkling water is sending a mild pain signal to your brain. And yet, millions of Americans reach for it every day. That’s because the brain processes this sensation very differently from, say, touching a hot stove. The low-grade stimulation is experienced as refreshing bite by most people, and only as true bitterness by those with heightened sensitivity.
Colder beverages can hold more dissolved CO₂, leading to a more pronounced fizzy sensation when consumed. The lower temperature also slightly numbs the mouth, enhancing the perception of tingling and bite. This is why a warm, flat sparkling water tastes significantly worse than an ice-cold one: you’re getting more of the acid flavor with less of the pleasant cooling effect to balance it out.

The Car4 Gene: Why Your DNA Decides How Bitter Sparkling Water Tastes
Here’s where things get genuinely personal. Not everyone experiences the bitterness of sparkling water equally, and the reason has everything to do with your genetics.
The gene that is linked to sour taste receptors is called Car4. Car4 works to create the bitterness you taste when you drink carbonated water by converting the CO₂ in the water to bicarbonate ions and protons. Those protons are what cause the bitterness in CO₂, which is then what causes the bitter taste in carbonated water.
Read More : Why Is Seagrams Ginger Ale Out Of Stock Updated 05/2026
Researchers theorize that the sensation of the bubbles in the carbonated water are what make the water taste bitter and not sour, while the carbonation itself does have an influence on how our bodies taste the CO₂.
Beyond the Car4 gene, the broader genetics of bitter taste perception play a massive role. The gene TAS2R38 encodes a specific bitter taste receptor on the tongue. Supertasters also tend to have a variant in the TAS2R38 gene that enables them to perceive the bitterness of chemical compounds similar to those in bitter vegetables. For the quarter of people who are supertasters, a coffee, or an IPA beer might be harshly bitter.
Are You a Supertaster?
About 25% of people are supertasters, 25% are non-tasters, and the other 50% are somewhere in the middle (medium tasters carry only one copy of the TAS2R38 gene, so fewer of those special bitter receptors and fewer taste buds than supertasters).
Women are more likely to be supertasters, at 35% of women and 15% of men. If you’re someone who finds IPAs overwhelming, can’t stand black coffee, or thinks grapefruit is aggressively harsh, there’s a real chance you’re a supertaster, and the bitterness you experience in sparkling water isn’t imaginary or exaggerated. It’s neurologically amplified.
For beer drinkers who love heavily hopped IPAs, this is ironic: the same TAS2R38 variant that makes bitter compounds pop in your IPA might also be exactly what makes a plain seltzer taste unpleasant. Conversely, those who find IPAs undrinkable often report that plain sparkling water is perfectly fine, even enjoyable.
Mineral Content: The Bitter Layer Beneath the Bubbles
Carbonic acid alone doesn’t explain the full range of bitterness experiences across different sparkling water brands. Mineral content is the other major variable, and it’s wildly different depending on what you’re drinking.
If sparkling mineral water contains a lot of sulfates, it can taste a bit medicine-like. When sodium is combined with chloride, they form sodium chloride that gives mineral water a salty taste. Also, a large quantity of magnesium can result in a bitter taste because it’s a base liquid. Bases taste bitter because they release hydroxide (OH) ions in water, which gives a bland taste, while acids release hydrogen (H) ions in the water, giving that sour taste.
This is why mineral-heavy European brands like Gerolsteiner are famously polarizing. Gerolsteiner is sharp, metallic, and bubbly. It is an assertive mineral water where you can taste the minerals. One taster described it simply: “really bitter, hated it.” Its high calcium and magnesium content means drinking it can be beneficial for health, but the same minerality that’s so good for you gives it that strong, intense flavor that turns some people away.
Here’s a breakdown of how popular brands compare in terms of bitterness, minerality, and carbonation intensity:
| Brand | Origin | Carbonation Level | Bitterness Profile | Best For |
|---|---|---|---|---|
| LaCroix | USA | Medium | Very mild, almost none | Everyday sipping, cocktail mixer |
| Topo Chico | Mexico | High | Clean, crisp, minimal bitter | Beer drinkers, margarita mixer |
| Perrier | France | Very high | Sharp, slight bitter edge | Aperitif, palate cleanser |
| San Pellegrino | Italy | Medium-high | Earthy, subtle mineral bitter | Food pairing, wine alternative |
| Gerolsteiner | Germany | High | Pronounced, metallic bitter | Adventurous mineral water fans |
| Bubly | USA | High | Mild, soda-like | Soda replacement crowd |
| Club Soda | Varies | High | Noticeably mineral, bitter | Cocktail mixing |
Perrier is known for its sharp effervescence and a distinct crispness on the palate. Tasting notes typically include a light and refreshing minerality with a subtle bitterness attributed to its higher levels of carbonation. San Pellegrino, on the other hand, provides a gentler fizz and is often described as having a smoother effervescence, finishing with a pleasing aftertaste that is less sharp.
For cocktail lovers especially, this matters enormously. Tonic water, for example, gets its signature bitterness not from carbonation but from quinine, a compound extracted from cinchona bark. What makes tonic water is the use of quinine, which gives it a bitter flavor. While the bitterness complements spirits well (say, gin or vodka), brands add sweeteners to combat the bitterness. Plain sparkling water has none of that quinine sharpness, but the carbonic acid bitterness is still there, which is why it behaves differently as a mixer than tonic does.
Temperature, Storage, and Why Your Warm Seltzer Tastes Terrible
If you’ve ever forgotten a can of sparkling water in a hot car and then tried to drink it, you know the result. It tastes flat, stale, faintly acrid, and nothing like the refreshing drink you were hoping for. This is chemistry, not coincidence.
High temperatures can also affect the taste and consistency of sparkling water. Leaving a can or bottle of sparkling water out in the heat can leave you with horrible-tasting water. Warm drinks lose their fizz because the loss of carbon dioxide increases with high temperatures. A decrease in the amount of dissolved gas can cause the drink to go flat and end up tasting bad. Moreover, the warmth or coldness of a beverage can affect our taste buds. Warm mineral water can increase your taste buds’ sensitivity to the taste of sodium and calcium.
Cold temperatures serve a dual purpose: they keep more CO₂ dissolved in solution (more fizz), and they mildly suppress some of the bitter receptor sensitivity in your mouth. This is why a glass of ice-cold Topo Chico with a lime tastes bright and refreshing, while the same Topo Chico at room temperature tastes like a flat, slightly medicinal puddle.
This principle applies directly to your drinking habits as a beer, wine, or cocktail drinker. A cold lager is served cold not just for tradition, but because warmth makes the hop bitterness more pronounced and the carbonation more aggressive and acidic. The same logic applies to sparkling water. Always serve it cold.
How Storage Affects Taste
Improper storage doesn’t just affect temperature. Exposure to light, especially in plastic bottles, can cause trace chemical interactions that alter flavor. Glass bottles tend to preserve the taste of sparkling water better, which is one reason why Perrier tends to be a bit fizzier from a glass bottle than the cheaper plastic bottles. If you’ve noticed your grocery store sparkling water tasting slightly off compared to the same brand served in a restaurant, bottle material is often the reason.
The Palate Paradox: Why Beer, Wine, and Cocktail Drinkers Are Uniquely Primed for Sparkling Water
There is an interesting relationship between experienced drinkers and the perception of sparkling water’s bitterness. If you regularly drink craft beer, especially hop-forward styles like IPAs and pale ales, you have likely trained your palate to interpret bitterness not as a flaw but as a signal of complexity and quality.
Regular exposure shifts tolerance. What initially tasted “bad” can become pleasant as sensory expectations adapt. The increased bitterness may be protective against alcohol intake in some individuals, while finer acuity may be advantageous for those who overcome the initial aversion and learn to like alcoholic beverages.
Read More : Who Makes Dr Thunder Soda Updated 05/2026
Wine drinkers may notice something similar. Tannins in red wine produce a dry, slightly bitter, astringent sensation on the tongue and gums. If you’ve developed an appreciation for a bold Cabernet Sauvignon or a structured Barolo, your brain has learned to file that bitterness under “sophisticated and desirable,” not “unpleasant.” Applying the same mental framework to sparkling water, especially mineral-rich varieties, can completely change how you experience it.
For cocktail enthusiasts, the connection is even more direct. Bartenders have long used sparkling water and club soda as mixers precisely because their slight bitterness counterbalances sweet elements in a cocktail. The CO₂ creates what mixologists call lift: it carries aromatic compounds upward, enhances the perception of lighter flavors, and adds a tactile brightness that still water simply cannot replicate. The tingling sensation associated with carbonated drinks is highly sought after and contributes to the mouthfeel and enjoyment of beverages, including beer, Champagne, and soft drinks.
Why Some People Never Get Used to It (And That’s Perfectly Okay)
Carbon dioxide, when dissolved in water, forms carbonic acid. This can give sparkling water a slightly acidic or bitter taste, which certain taste buds are more sensitive to. Genetic variations play a significant role in taste perception. Some people have heightened sensitivity to bitter flavors, making the mild acidity of sparkling water more pronounced.
Beyond genetics, there’s a strong psychological component. People who associate sparkling water with flat soda may carry a preconceived notion of staleness, influencing their dislike. Taste preferences are not solely determined by biology; psychology and cultural factors also play a role.
If you grew up drinking primarily sweet sodas like Coca-Cola or Mountain Dew, the absence of sweetness in plain sparkling water can feel jarring. The bitterness isn’t stronger, it just has nothing to hide behind. This is the same reason many new beer drinkers find their first IPA overwhelming: without context or contrast, the bitter compounds dominate. Over time, the brain recalibrates.
How to Make Sparkling Water Less Bitter: Practical Tips
If you enjoy the hydration and the fizz but find the bitterness of plain sparkling water off-putting, there are several straightforward ways to dial it back.
Add citrus. A squeeze of fresh lemon, lime, or orange juice introduces natural acidity that actually complements the carbonic acid instead of clashing with it. Your brain interprets the combined flavors as “sour and bright” rather than “bitter and flat.” This is why sparkling water with lime is a staple alongside tequila shots. One easy combination is a shot of tequila, a glass of sparkling water, and lemon or lime juice. It’s very refreshing.
Choose the right brand for your palate. Not all sparkling waters are equally bitter. Due to its balanced content of calcium, magnesium, and bicarbonate and comparatively small amounts of sodium and chloride, Gerolsteiner Mineral Water tastes neither salty, sour nor bitter, but pleasantly natural and neutral, according to the brand. If heavily mineralized waters are too aggressive, a lightly mineralized option like LaCroix or a premium American brand like Saratoga Springs may offer a cleaner, less bitter experience.
Serve it very cold. As discussed, cold temperatures suppress some bitter receptor sensitivity and hold more CO₂ in solution, giving you more fizz and less lingering acid flavor.
Use it as a mixer. When carbonated water is combined with other flavors and drinks, the bitter taste is not noticeable. A splash of fruit juice, a few dashes of bitters (yes, ironically), or a measure of spirits transforms the bitterness into background texture. This is exactly why sparkling water has become such a staple at the bar.
Let it breathe briefly. CO₂ will naturally escape from sparkling water if you let it sit in a glass. You can also make it taste less bitter by adding sweet fruit juice or syrup to it. Apart from adding natural sugar to give it a sweet taste, it also dilutes the amount of CO₂ in the sparkling water. Pouring into a glass and waiting thirty seconds allows some CO₂ to escape, softening the bite considerably, which is how it’s commonly served in fine dining settings.
The Sparkling Water Boom: Why More Americans Are Drinking It Than Ever
The bitterness of sparkling water clearly isn’t stopping anyone. As of Q1 2024, the global sparkling water market is valued at approximately $63.5 billion. U.S. soda consumption dropped 5% in 2023, while sparkling water rose 9.8%.
Health-behavior surveys from 2024 reveal stronger label scrutiny among younger consumers, further driving the shift away from sugary sodas. This trend is particularly prominent among Millennials and Gen Z, who together represented over 42% of the United States population in 2024.
The US Sparkling Water Market was valued at USD 5.5 billion in 2024 and is projected to reach USD 12.0 billion by 2034, growing at a CAGR of 8.0%. The trend of mixology and craft cocktails has also led to a rise in the use of sparkling water as a mixer, further driving demand in the food service sector.
For American adults who drink alcohol socially, sparkling water has taken on a new social role. It’s the drink between drinks, the palate cleanser, the designated driver’s go-to, and increasingly, the sophisticated choice at bars where a Topo Chico with lime carries as much cachet as a craft cocktail.
A Note on Club Soda vs. Sparkling Water vs. Seltzer (The Differences Actually Matter)
These terms are often used interchangeably, but they produce noticeably different flavor experiences:
- Seltzer: Plain water carbonated with CO₂. No added minerals. The bitterness is purely from carbonic acid. Cleanest and most neutral. LaCroix and Bubly fall here.
- Club soda: Carbonated water with added minerals, typically sodium bicarbonate, potassium sulfate, or disodium phosphate. The addition of minerals make this similar to sparkling mineral water, and some may find club soda to have a savory taste. It’s the go-to for cocktail mixing because minerals add complexity without flavor.
- Sparkling mineral water: Water from a natural spring source, containing naturally occurring minerals. CO₂ is often added back after bottling. The bitterness profile is entirely shaped by the mineral composition of the source. San Pellegrino, Perrier, and Gerolsteiner fall here.
- Tonic water: Carbonated water with added quinine and sugar. The bitterness is dramatically more pronounced than any of the above, which is why it stands up so well against bold spirits like gin.
Understanding which is which is essential if you’re using sparkling water in cocktails, as each brings a different flavor foundation to the glass.
Conclusion
The bitterness of sparkling water is not a defect in your drink or your palate. It’s a conversation between chemistry and biology, between carbonic acid and a trigeminal nerve that has been quietly guarding your mouth since long before sparkling water was sold in cans. Your DNA influences how loud that conversation gets. Your drinking history shapes whether you hear it as music or noise.
The next time you’re standing at the bar, deciding between a round of drinks and a glass of sparkling water, know this: the slight bitterness in that glass is the same basic phenomenon that makes Champagne sparkle, that gives a good witbier its crispness, and that makes a perfectly made gin and tonic so electrically alive. You’ve been appreciating carbonation-driven bitterness your whole drinking life. Sparkling water is just the unmasked version of it.
Sources: https://chesbrewco.com
Category: Drink